Steve Jacobsen on Materials in Extreme Environments
Listen to the podcast here or wherever you get your podcasts.
Almost all the material that makes up the Earth and other planets exists under extremes of pressure and temperature quite unlike those we inhabit on the surface of the Earth. In the podcast, Steve Jacobsen discusses how he mimics such conditions in the lab so as to learn how rocks and minerals behave under such alien conditions. The discussion focuses on three extreme environments: the core-mantle boundary (for which he uses the massive Z Machine, designed for thermonuclear fusion experiments), the mantle transition zone, and the surface of the Moon.
Jacobsen is a Professor of Geological Sciences at the University of Colorado Boulder. The image shows him in his optical spectroscopy lab, where extreme conditions found throughout the solar system are re-created.
Podcast Illustrations
Images Courtesy of Steve Jacobsen unless otherwise noted.
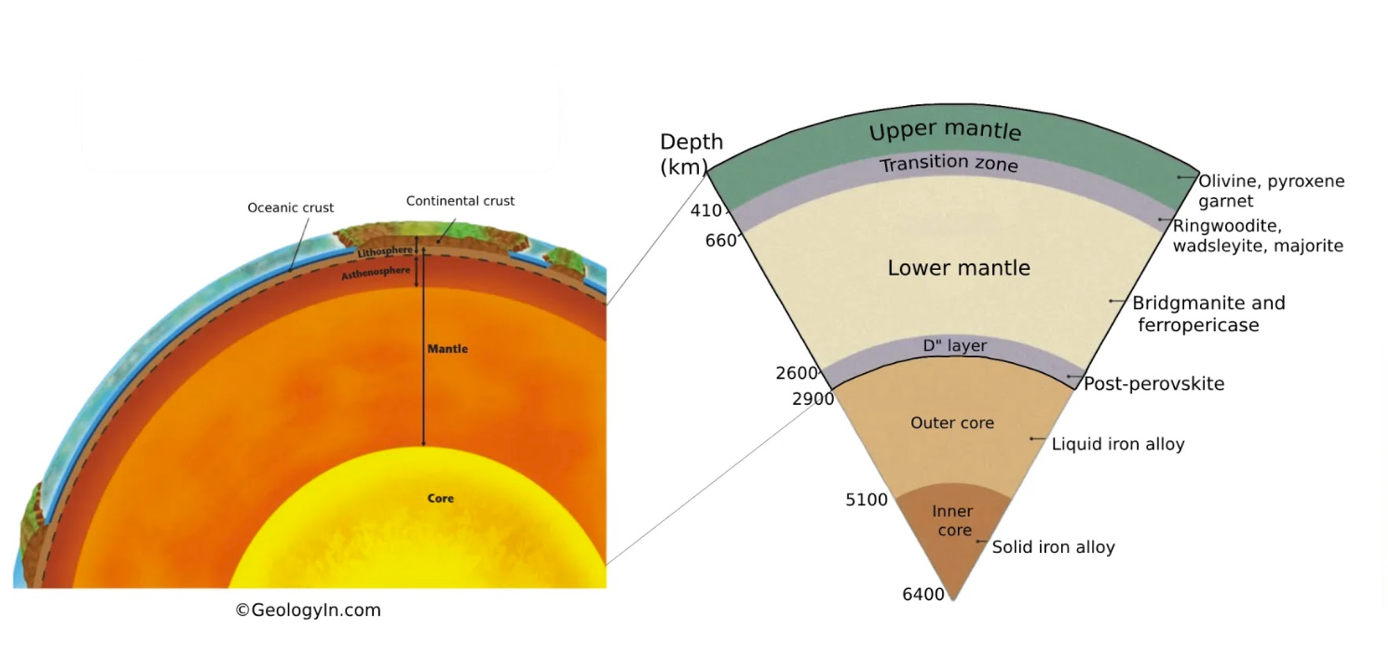
Diagrammatic cross-section of the earth showing the core-mantle boundary at a depth of 2,900 km and the mantle transition zone between the seismic-wave speed discontinuities at 410-km depth and 660-km depth.
Copyright GeologyIn.com
Core-Mantle Boundary
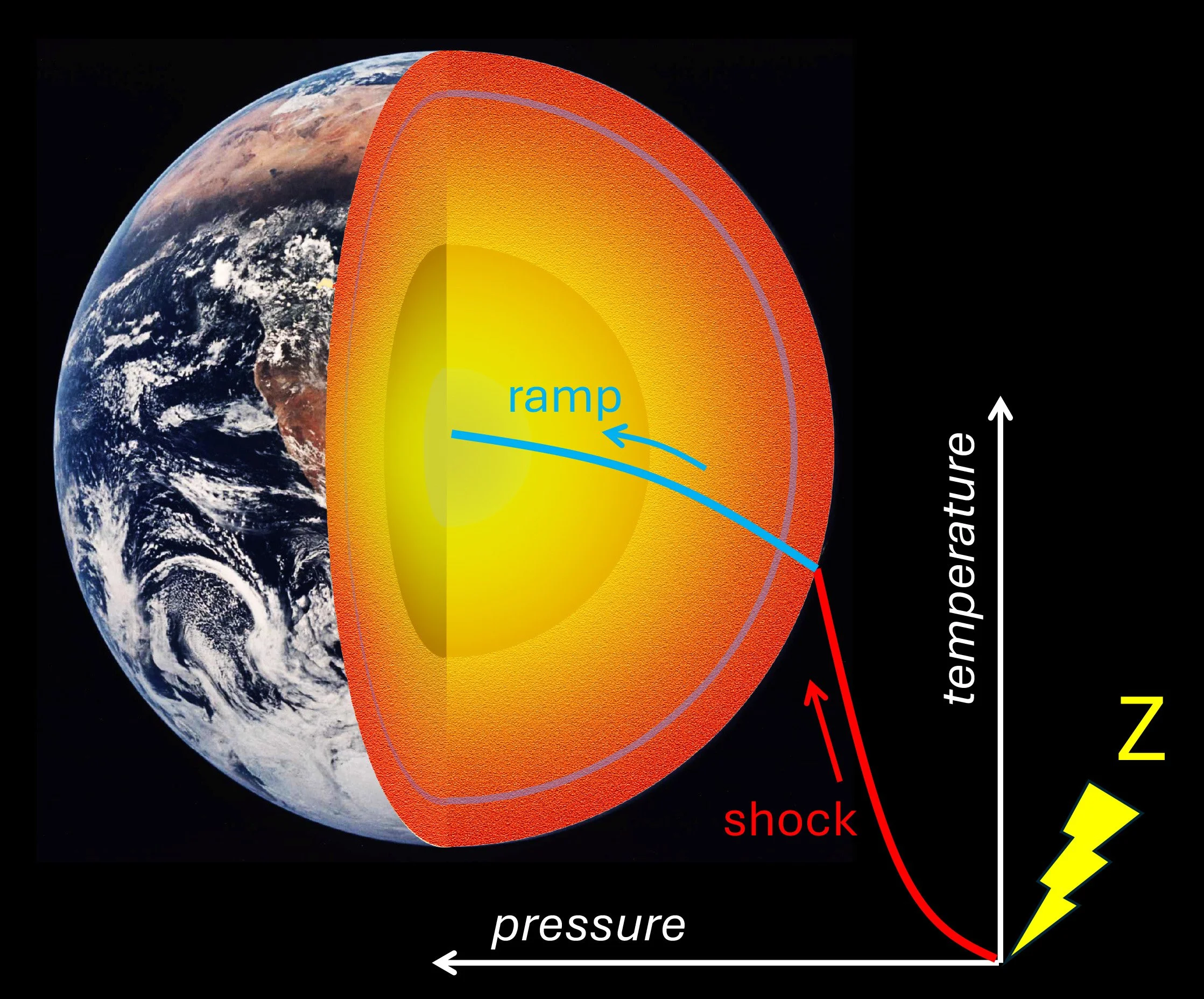
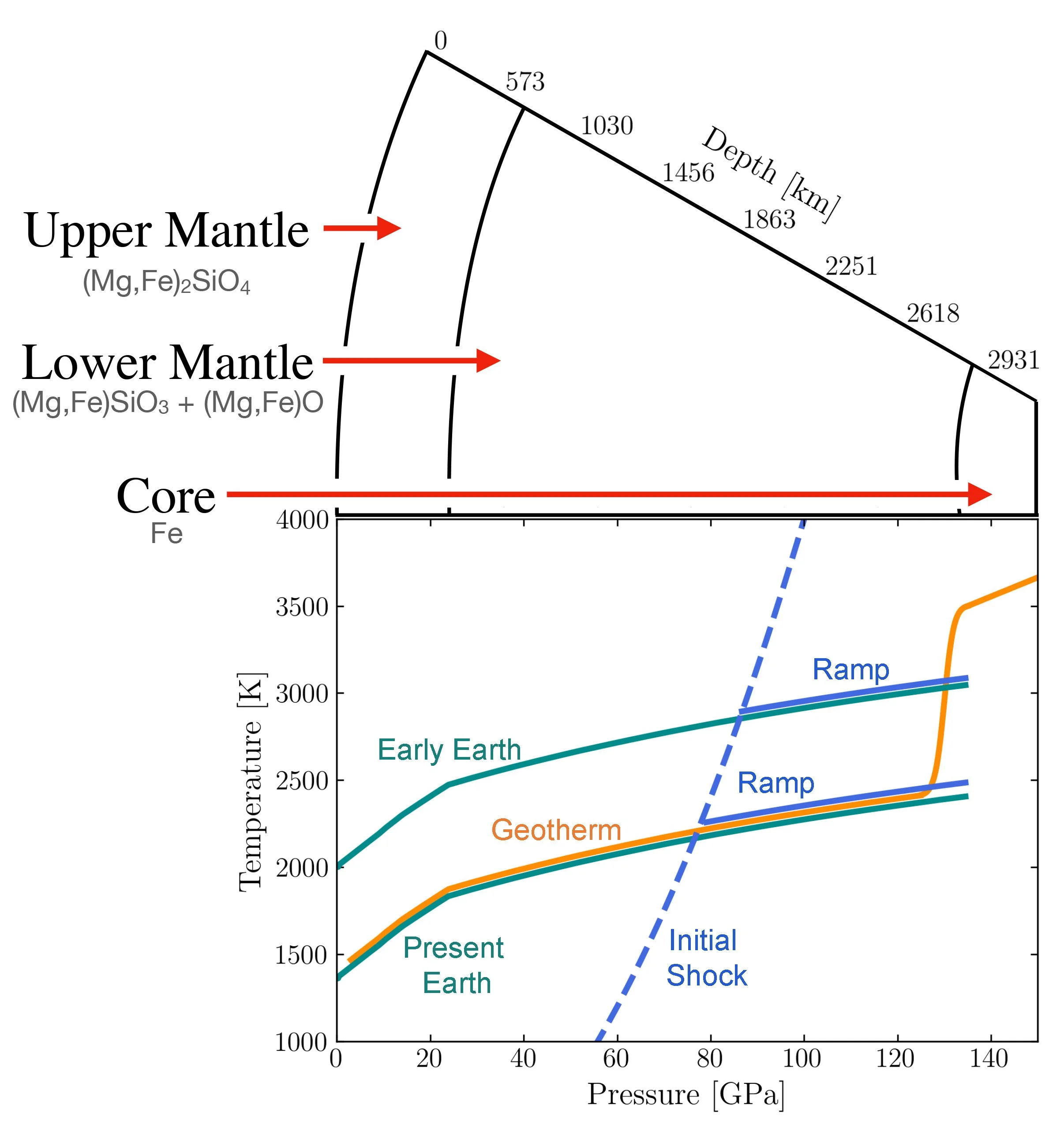
In order to reach the temperature and pressure conditions that prevail at the core-mantle boundary, the sample is subjected to a two-stage process: an initial shock to heat the sample, followed by a subsequent shockless “ramp” compression that increases the pressure without significantly further heating. If the sample was just shocked to reach the required pressure, it would be accompanied by heating to a temperature much higher than the target 4,000K temperature. This is indicated by the dotted blue line in the diagram shown below.
Schematic diagram showing how temperature and pressure increase with depth in the Earth. Superposed on the diagram in blue are the temperature-pressure paths taken by samples in the experiments Jacobsen describes in the podcast. The dotted line shows how the temperature and pressure states increase in the sample if it is simply shocked, equivalent to being hit by a hammer. But to reproduce the conditions in the mantle, the experiments depart from the shock curve as shown by the solid blue lines and are intended to follow temperatures and pressures that occur along the Earth’s geotherm, shown in green for both the present Earth and a hotter, early Earth for comparison.
The Z Machine at Sandia National Laboratories, Albuquerque, New Mexico. In the podcast, Jacobsen describes how he uses this machine to study the properties of mantle material at the pressures and temperatures that prevail at the core-mantle boundary. The Z machine is 33 metres in diameter and is the most powerful pulsed-power facility in the world. The concentric rings of metal modules visible in the image are banks of capacitors that store huge amounts of electrical energy. When fired, all of this energy is discharged simultaneously inward along transmission lines toward the small central hub, where a tiny target sample sits. There, the 20-mega-amp pulse — lasting a few nanoseconds — generates the desired pressures and temperatures in the sample. The yellow safety railings, walkways, and blue water-cooling pipes give a sense of the machine's industrial scale, yet the science happens in a target no larger than a thimble at the very center.
Sandia National Laboratories
The Z Machine during a discharge, captured in a long-exposure photograph. The impressive branching arcs and blue-white glow are produced by the intense electromagnetic fields ionising the surrounding air — essentially, energy leaking from the machine during the pulse. The actual experiment is hidden from view, taking place inside a sealed target chamber at the center.
Sandia National Laboratories
Steve Jacobsen (right) and a colleague examine a target assembly for the Z Machine at Sandia National Laboratories. The precision-machined disc carries the tiny mineral sample at its center — the thimble-sized target where the full power of the machine's converging electrical pulse will be focused. This is the hardware that bridges the 33-metre-wide ring of capacitors seen in the above images and the millimetre-scale crystal samples shown below.
Sandia National Laboratories
Diamond anvil cell. In the podcast, Jacobsen also describes using a diamond anvil cell to compress samples to core-mantle boundary pressures but maintaining the sample at room temperature. These experiments enable him and his team to use X-ray diffraction to determine the structure of the material at high pressure and also to study the sample’s optical properties.
Crystals of ferropericlase with varying iron content. From left to right: pure MgO (periclase), transparent and colorless; 5% iron, amber; 20% iron, nearly black; and pure FeO (wüstite), fully opaque. In the podcast, Jacobsen describes how increasing the iron content of ferropericlase simultaneously raises its density and lowers its shear-wave velocity — a combination that could explain the puzzling seismic signature of the ultra-low velocity zones at the base of the mantle, where material is both anomalously dense and anomalously slow. His experiments on the Z Machine aim to measure these properties across a range of iron contents at core–mantle boundary pressures, to see whether iron-enriched ferropericlase can match what seismologists observe.
Mantle Transition Zone
Wadsleyite crystals. These deep green crystals are the high-pressure form of olivine that is thought to be the dominant mineral in the mantle transition zone, between about 410 and 520 km depth. Wadsleyite is of particular interest because its crystal structure can incorporate small amounts of water as hydroxyl groups — making the transition zone a potentially vast reservoir of water within the Earth. These crystals were synthesized in the laboratory at high pressure; wadsleyite does not survive the journey to the surface intact, as it reverts to olivine when pressure drops.
A crystal of ringwoodite. This is the high-pressure polymorph of olivine that is stable between about 520 and 660 km depth in the mantle transition zone. In the podcast, Jacobsen points out ringwoodite's remarkably large capacity to incorporate the chemical components of water as hydrogen defects in its crystal structure. This property makes the transition zone a potentially enormous reservoir of water within the Earth. Jacobsen suggests that the transition zone may act as a kind of sponge, absorbing water and other incompatible elements from ascending melts — an idea developed by Bercovici and Karato in their "transition zone water filter" model. He goes further, speculating that a planet may need to be large enough to sustain the pressures required for ringwoodite to exist before it can hold liquid water on its surface — noting that Mars, which barely reaches these pressures, has no transition zone and no surface water.
Image by Jasperox CC BY 3.0
The Surface of the Moon
Artist's rendering of ICON's Project Olympus concept for 3D-printing a landing pad on the lunar surface using local regolith. In the podcast, Jacobsen describes how his group is helping to develop the materials science behind this kind of additive manufacturing — using lasers to melt simulated lunar soil into solid building material, one layer at a time.
ICON
A cross-section through a 3D-printed brick made from lunar regolith simulant by laser powder bed fusion. The layering is clearly visible — each layer represents a single pass of the laser melting a bed of simulated lunar soil, which then solidifies before the next layer is added. In the podcast, Jacobsen describes the material as having roughly the composition of a mixture of basalt and anorthosite. The porosity and bubbles result in part from volatile elements such as sodium and potassium boiling off during melting. A key challenge Jacobsen discusses is that real lunar regolith — modified by 4.5 billion years of space weathering and coated with nanoscale iron particles — may behave quite differently from the simulants used in the lab, particularly in how it absorbs laser energy.
Tirzah Abbott / Northwestern University
Jacobsen's research on materials in extreme environments is supported in part by the US Department of Energy (DOE), National Nuclear Security Administration (NNSA) through the Chicago/DOE Alliance Center (CDAC), and by the US National Science Foundation (NSF), and by NASA.